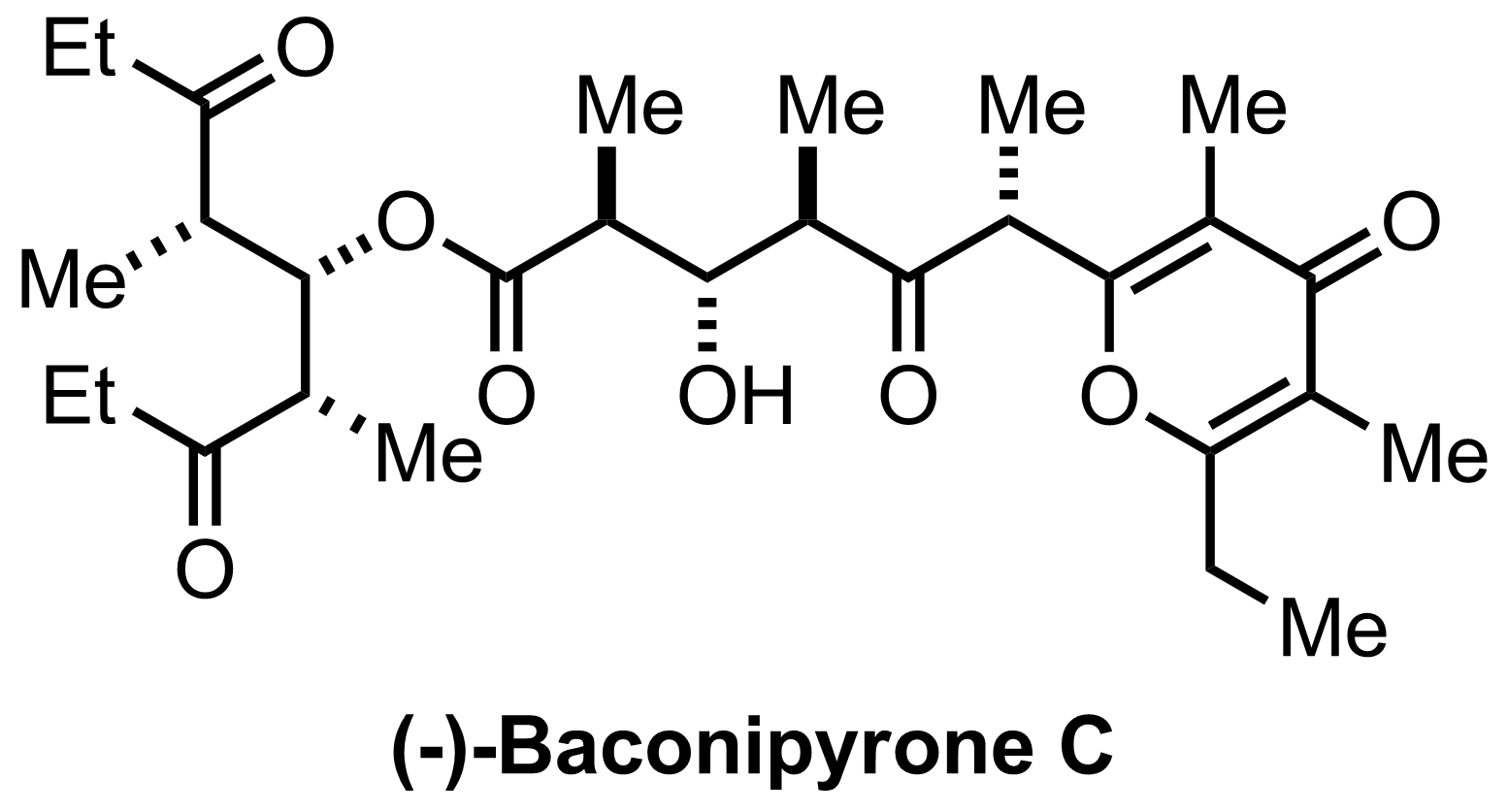
Synthesis of Baconipyrone C
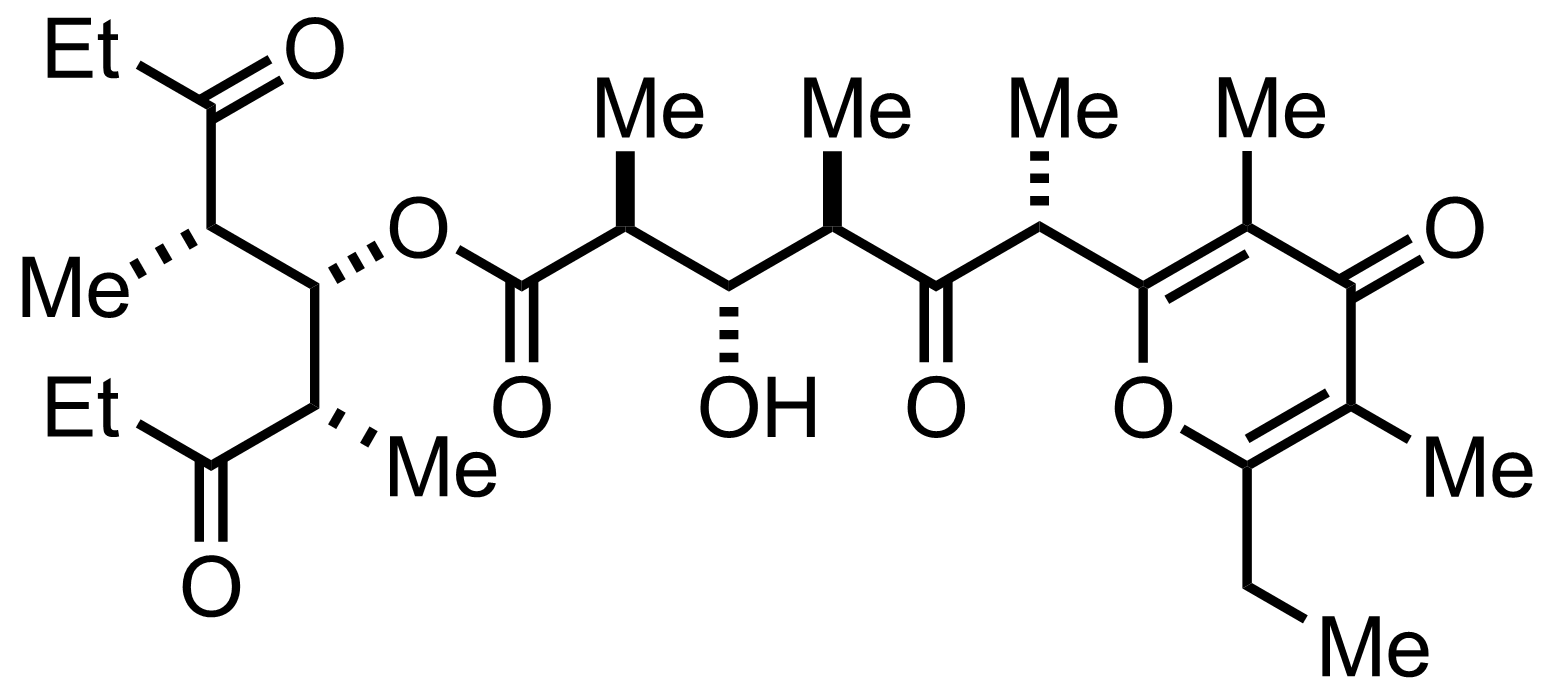
C29H44O8
| Principal investigator | Ian Paterson |
|---|---|
| Publication year | 2000 |
| Synthesis type | Total |
| Number of steps | 23 (3 parts) |
| References |
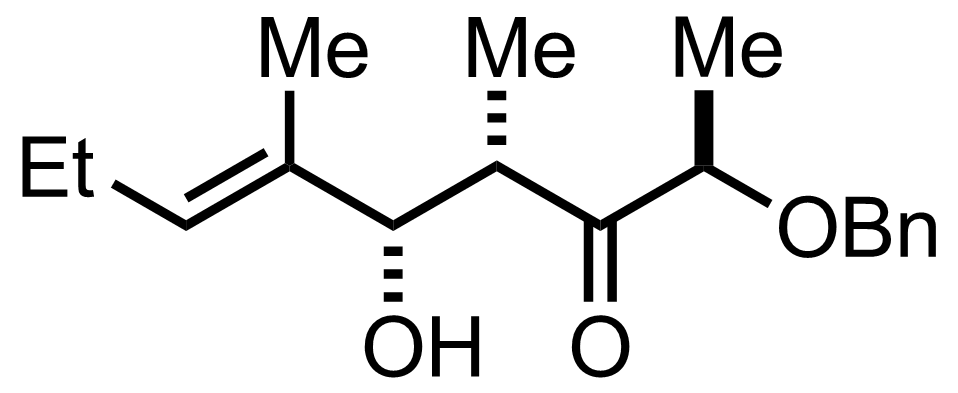
Part 1 of 3
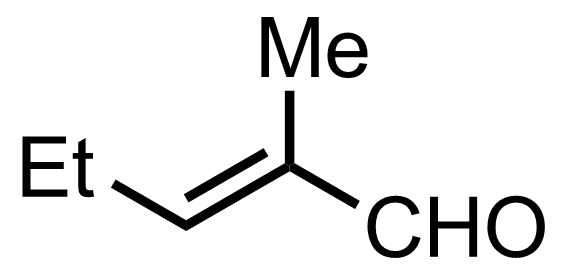 +
+
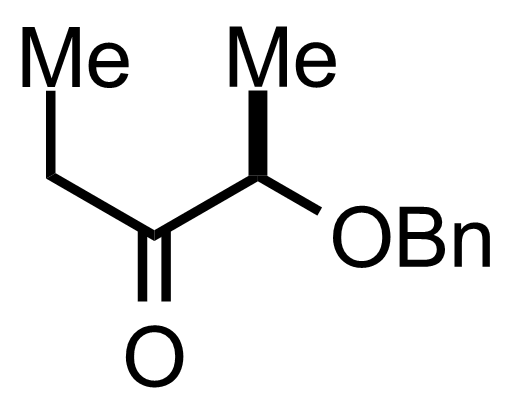
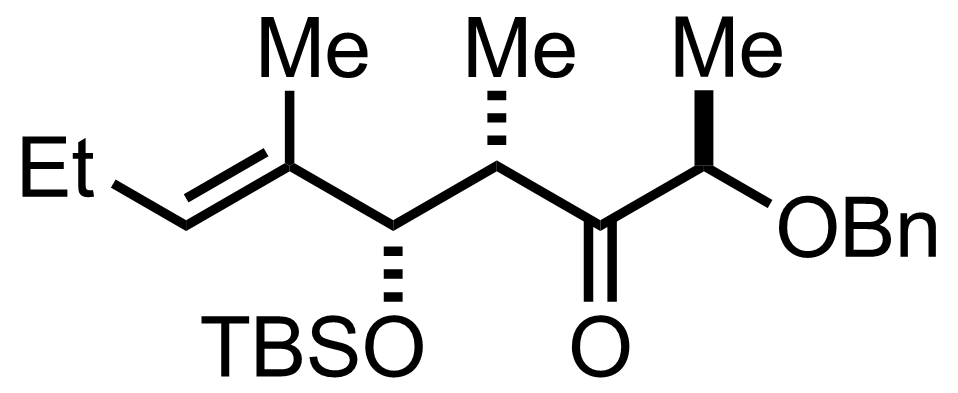
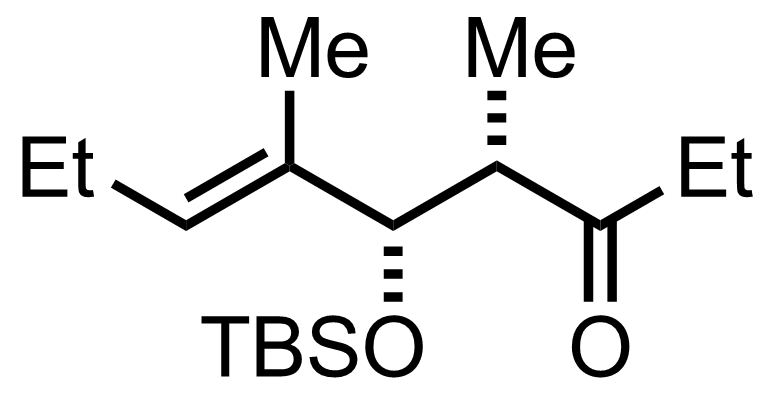

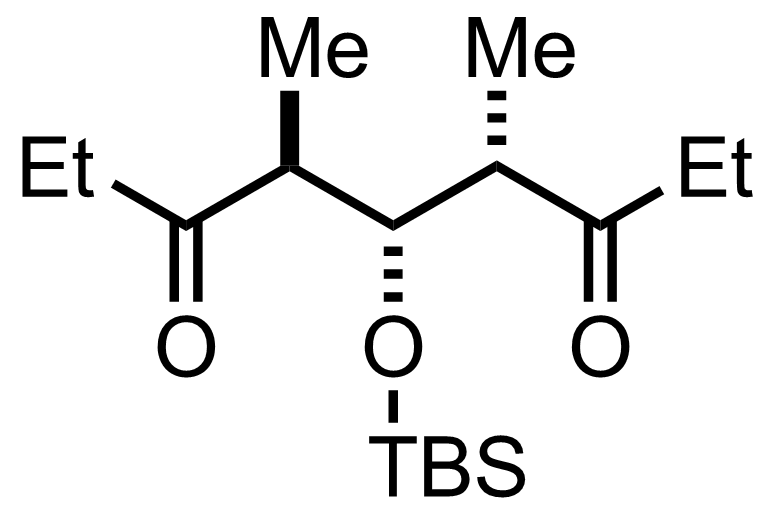

 +
+


TBSOTf,
2,6-Lutidine
-78 °C, 99%

SmI2
THF
0 °C, 94%

- Thexylborane
- H2O2 , NaOH
H2O, THF
RT to 40 °C, 17 h, 59%


Pyr.HF
THF
0 °C, 60 min, 97%

Part 2 of 3
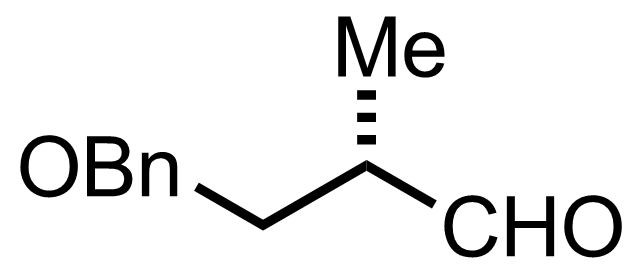 +
+
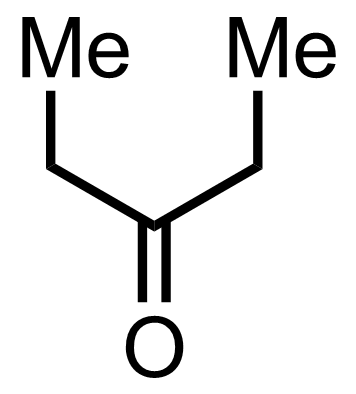
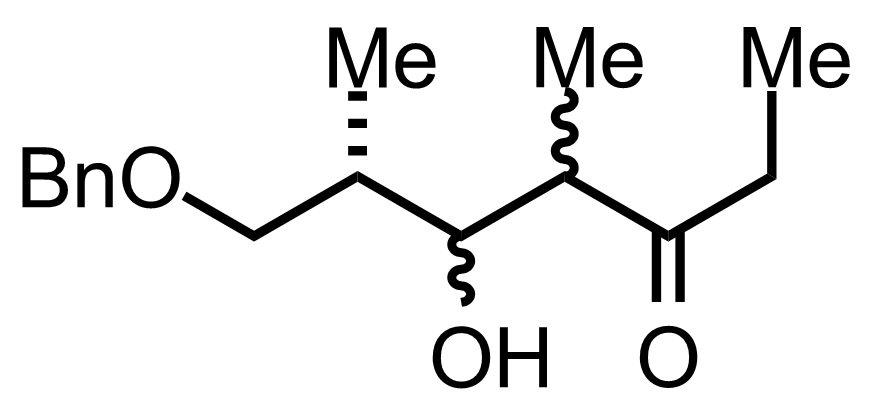
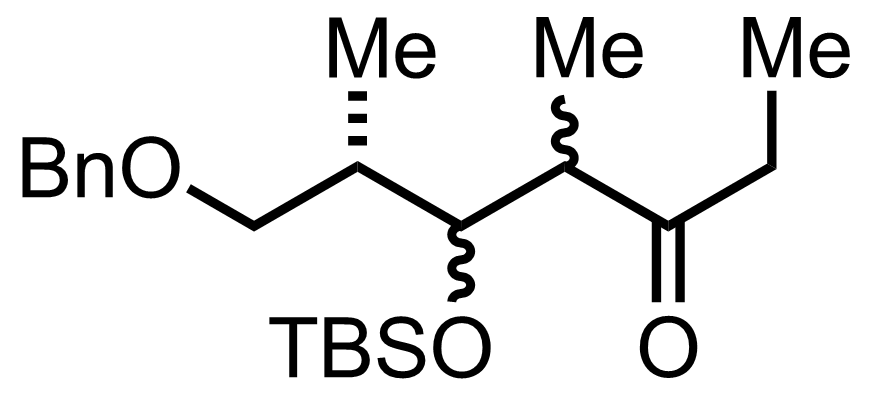
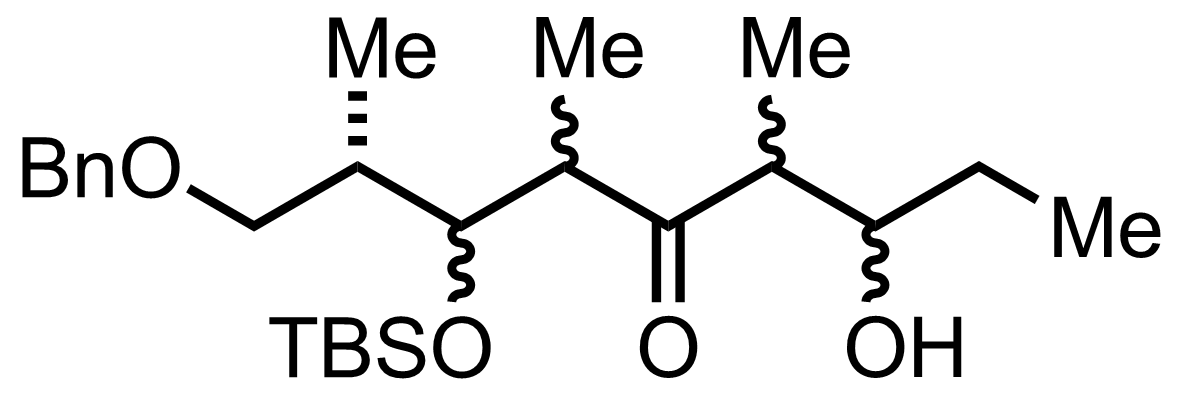

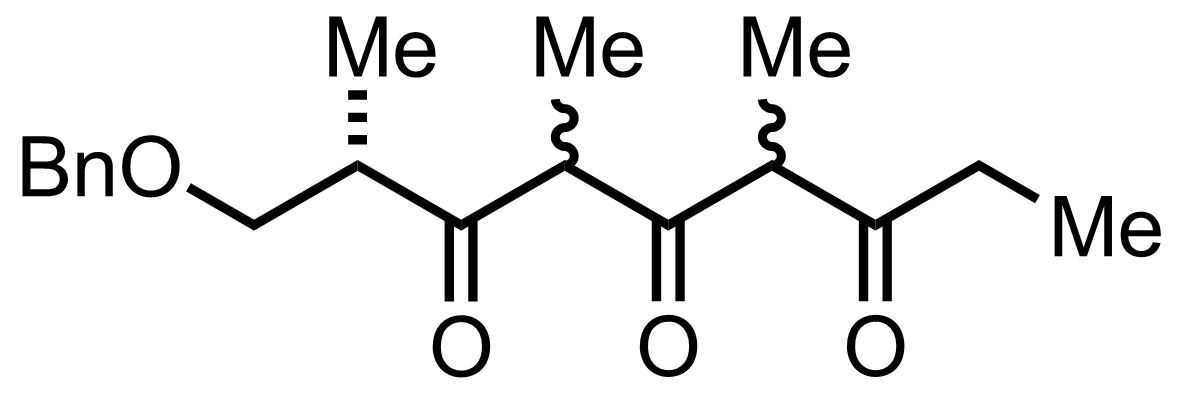

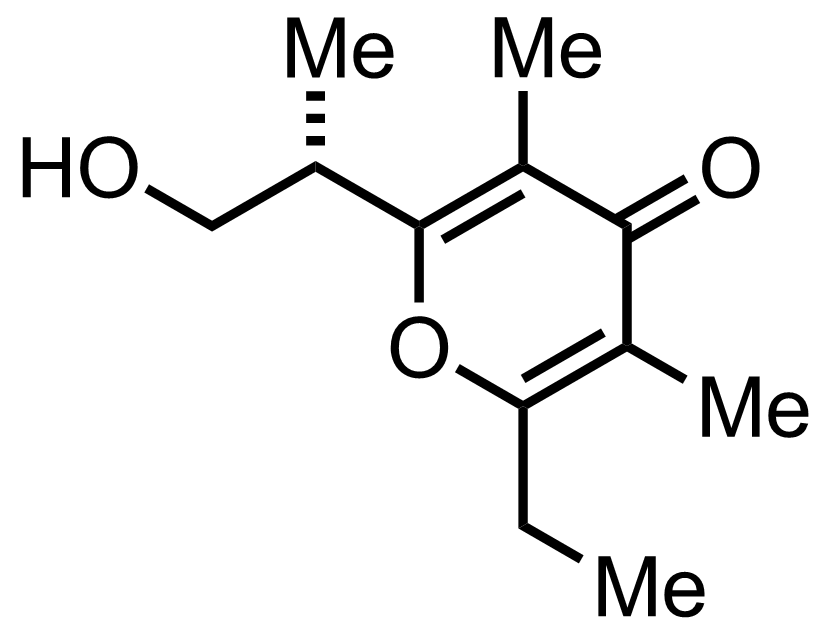
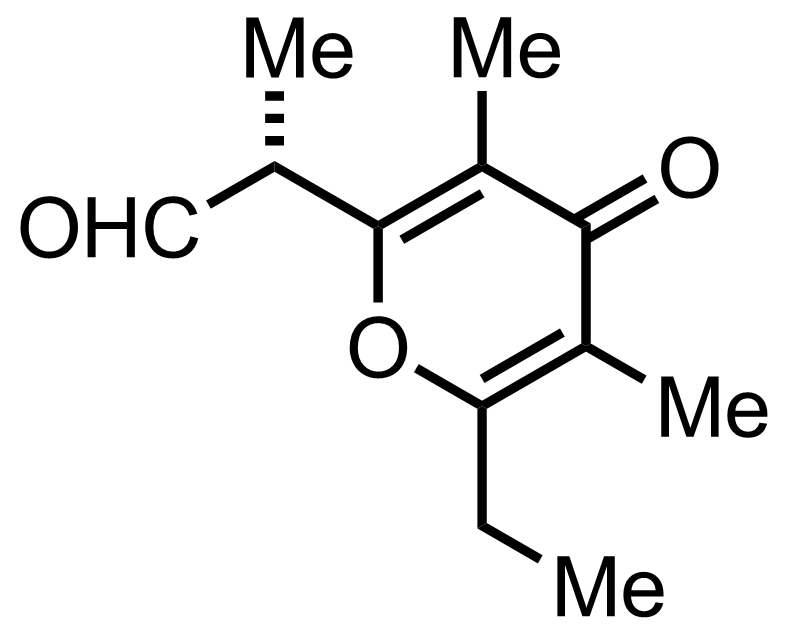
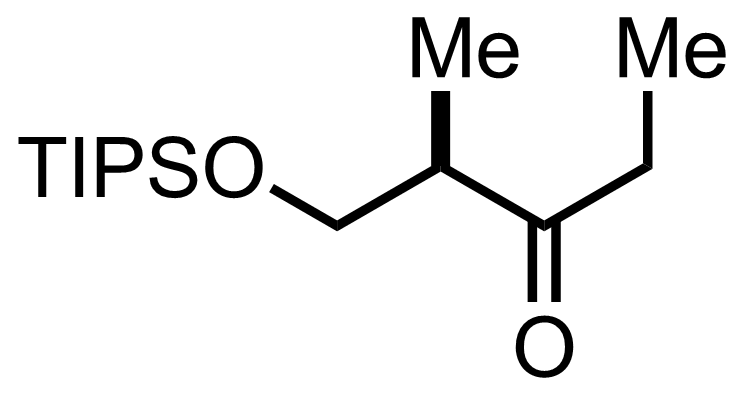

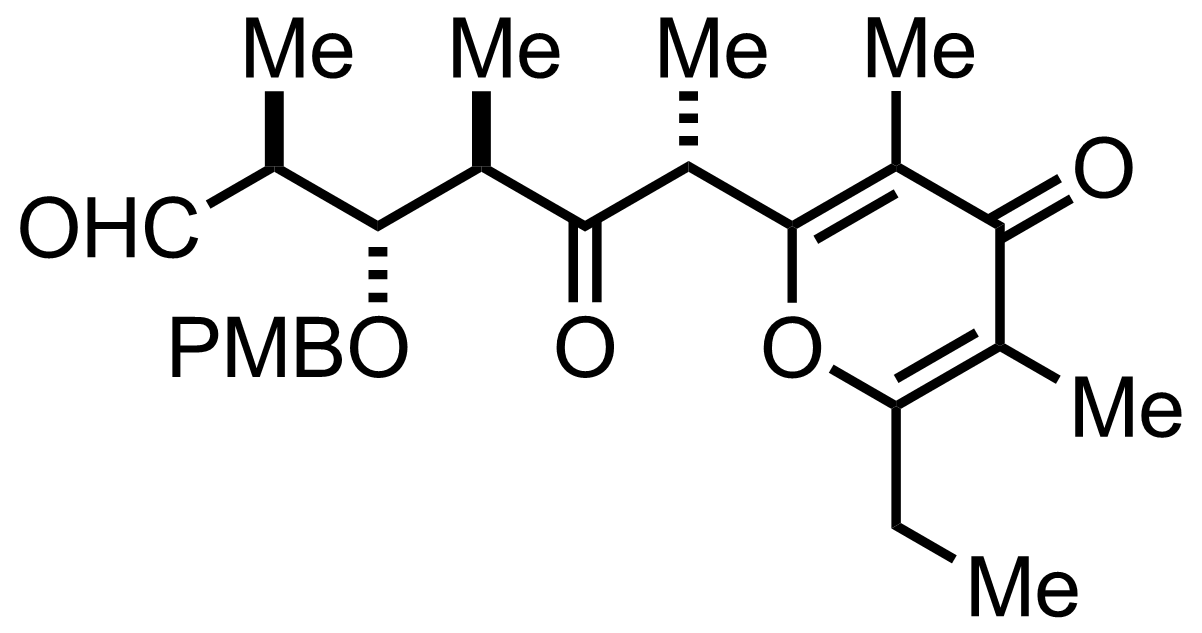
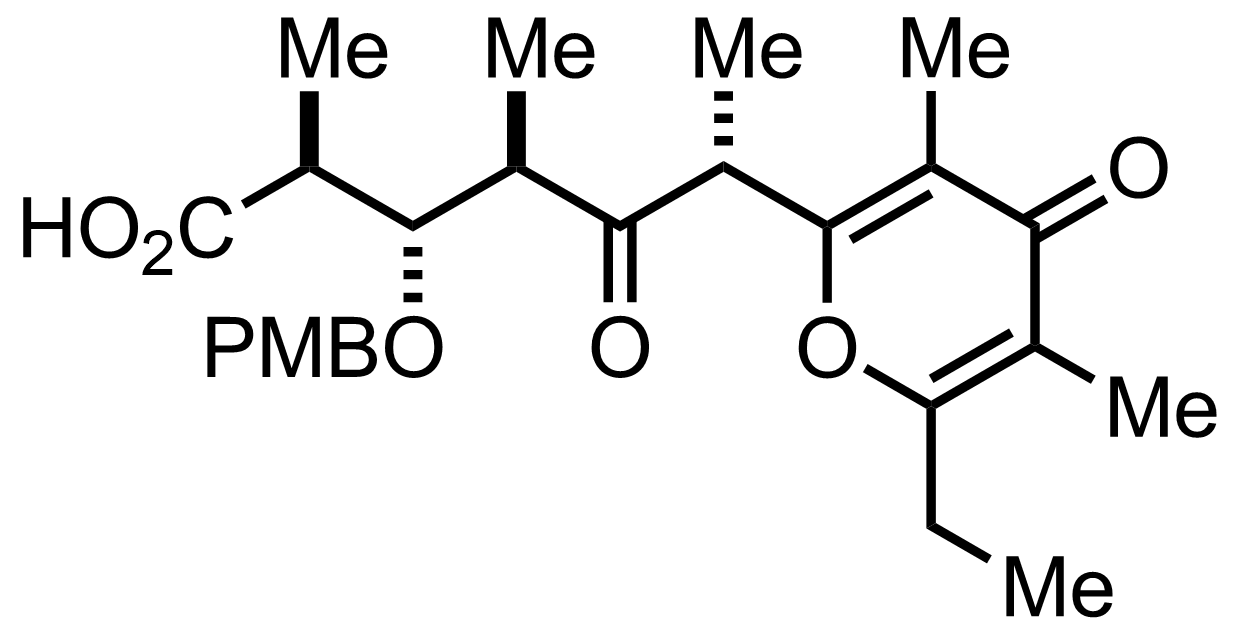
 +
+


2,6-Lutidine,
TBSOTf
CH2Cl2
-78 °C, 2 h


HF
MeCN
20 °C, 2 h


Ph3P
CCl4, THF
RT, 3 d, 88%

H2,
Pd/C
1 atm
EtOH
RT, 8 h, 95%



Sn(OTf)2,
Et3N
CH2Cl2
-78 °C, 4.5 h, 74%
See the Aldol Addition
"Also isolated were 6% of other diastereomers"


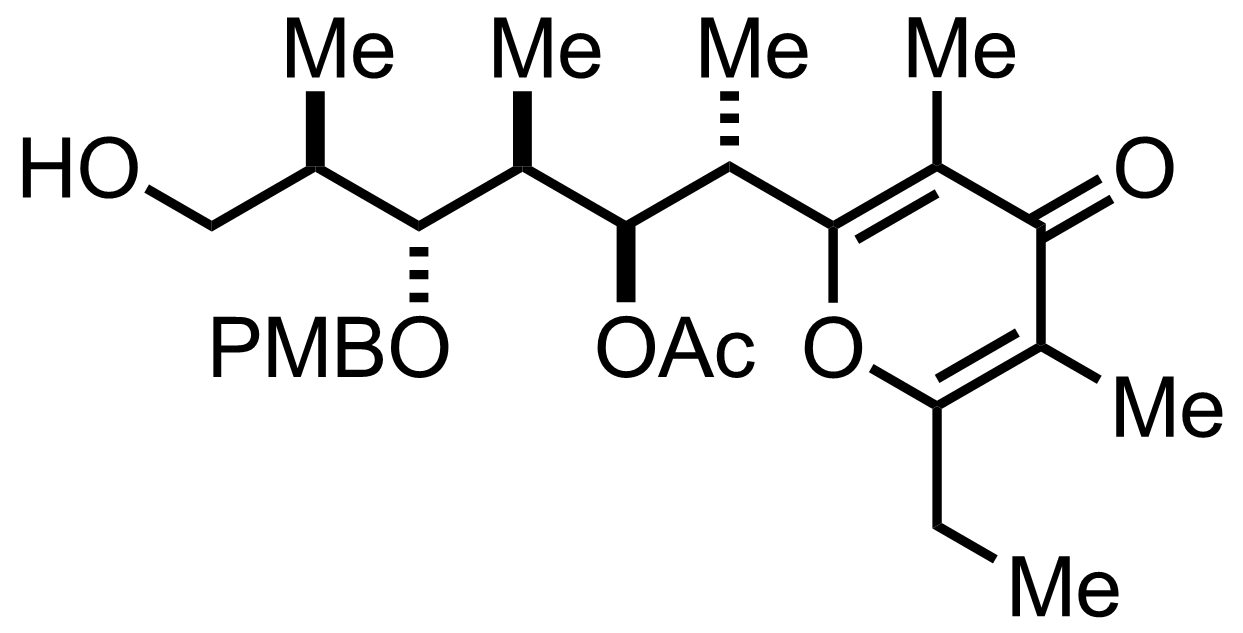
TfOH,
PMBOC(=NH)CCl3
Et2O
94%

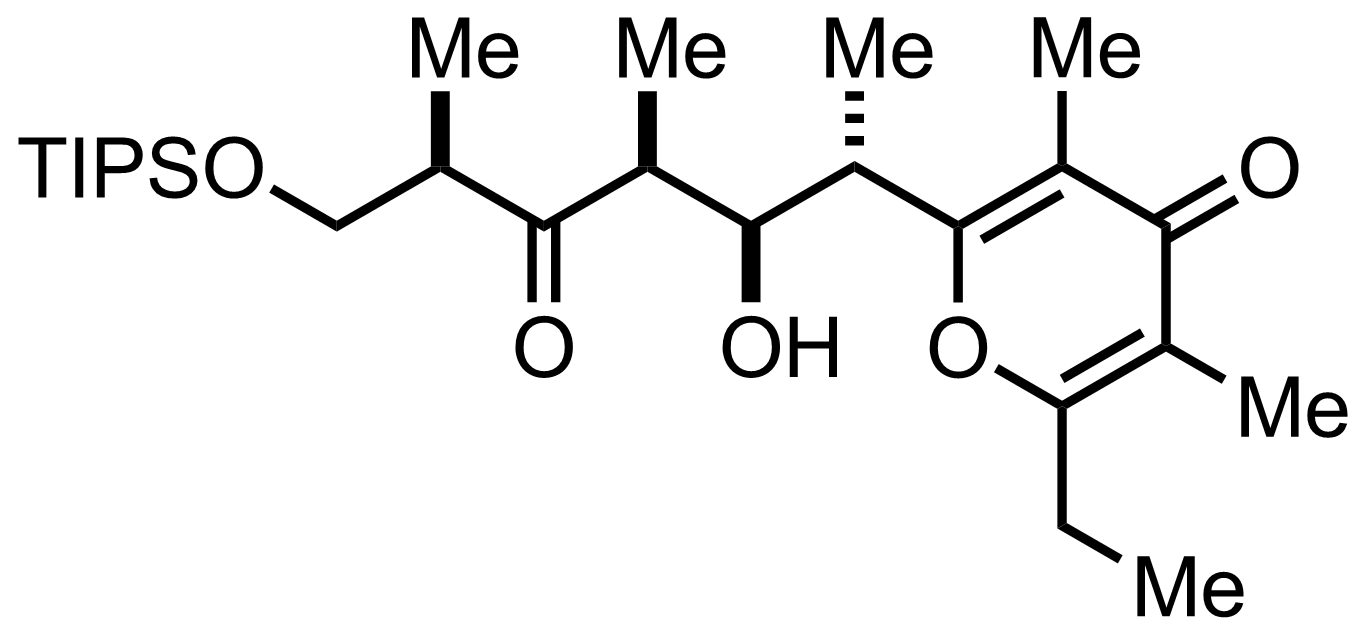
n-Bu4N+ F-
THF
87%

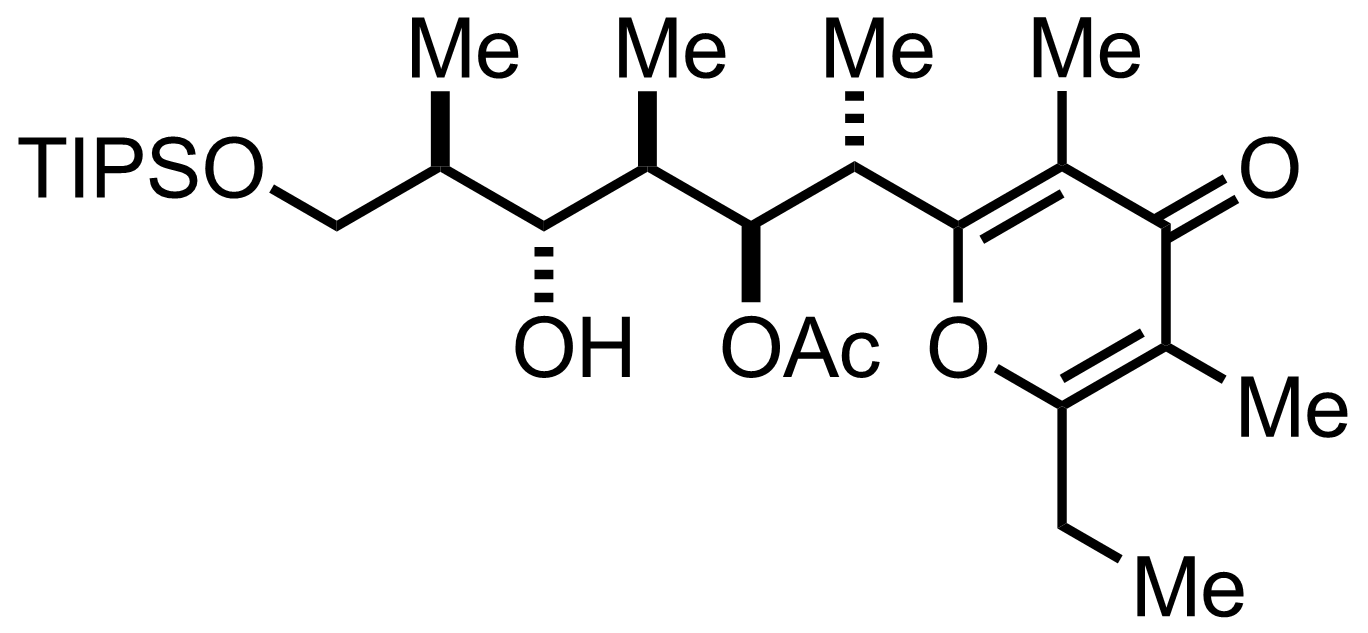
K2CO3
MeOH
60 °C, 97%


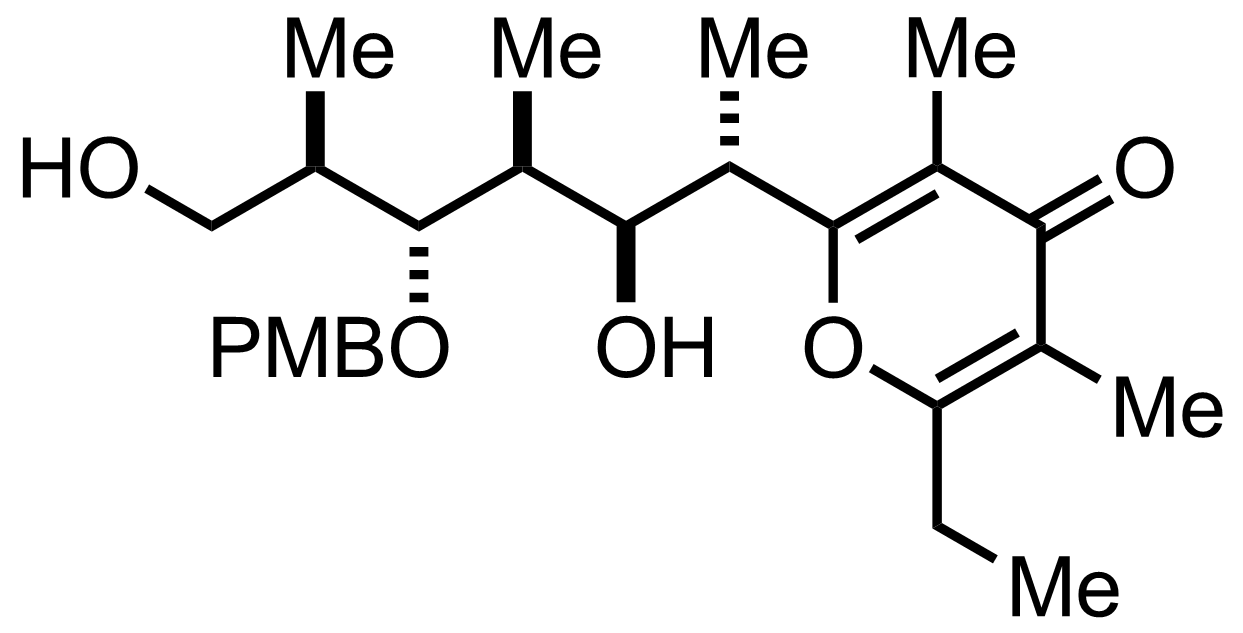
Na2HPO4,
NaClO2,
2-Methyl-2-butene
H2O, t-BuOH
RT, 1 h, 96% (2 steps)
See the Pinnick-Lindgren Oxidation

Part 3 of 3
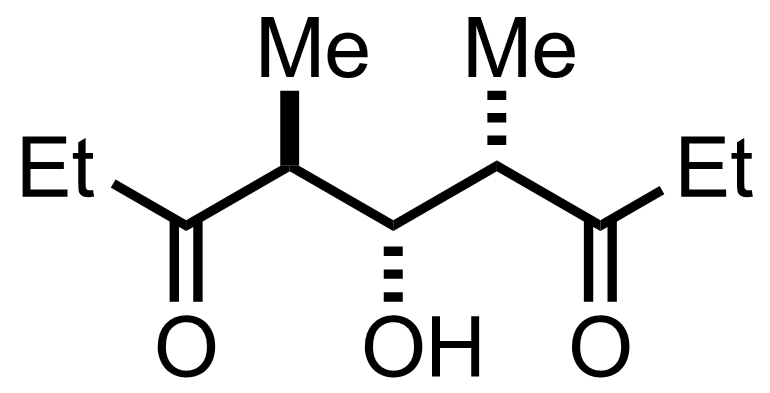 +
+
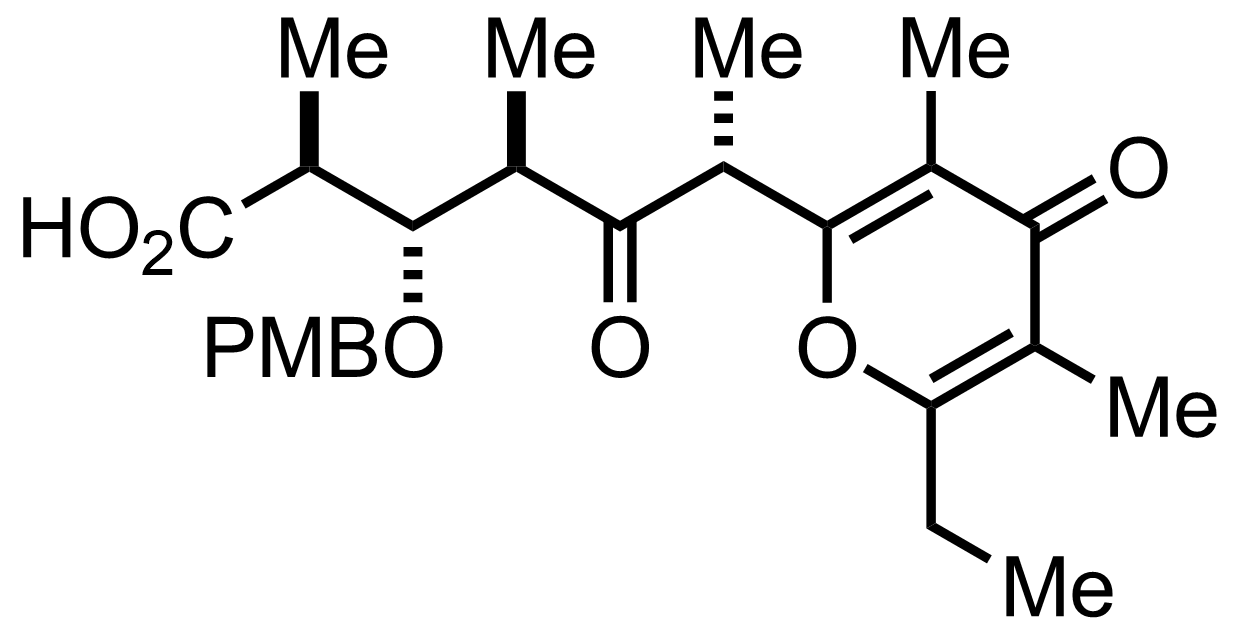
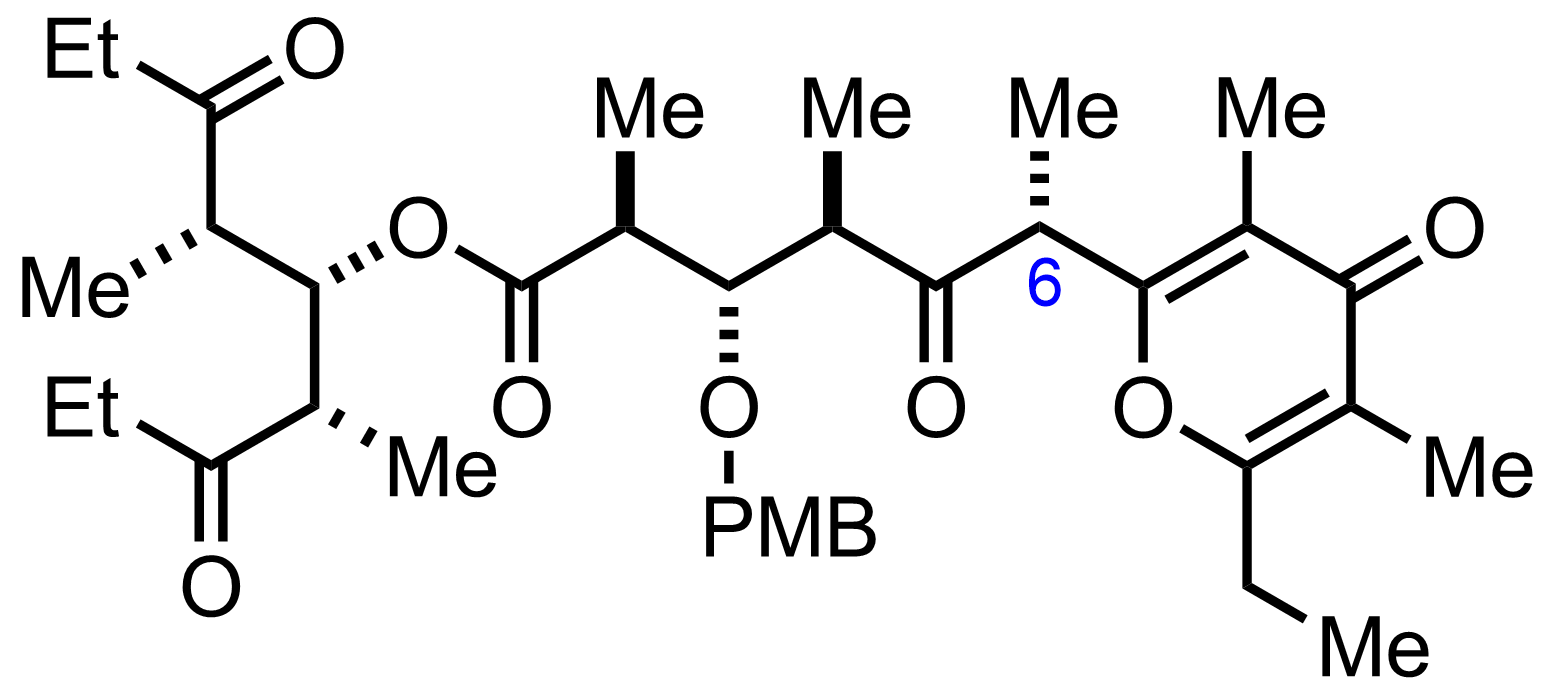
 +
+

DMAP,
Et3N,
2,4,6-Trichlorobenzoyl chloride
PhMe
-78 to 0 °C, 10 min, 73%
See the Yamaguchi Esterification
"selectivity 91:09"

DDQ
pH=7 buffer
CH2Cl2
RT, 1 h, 67%
"Also isolated was 7% of C6-epimer"

