Synthesis of Spiroindimicin B
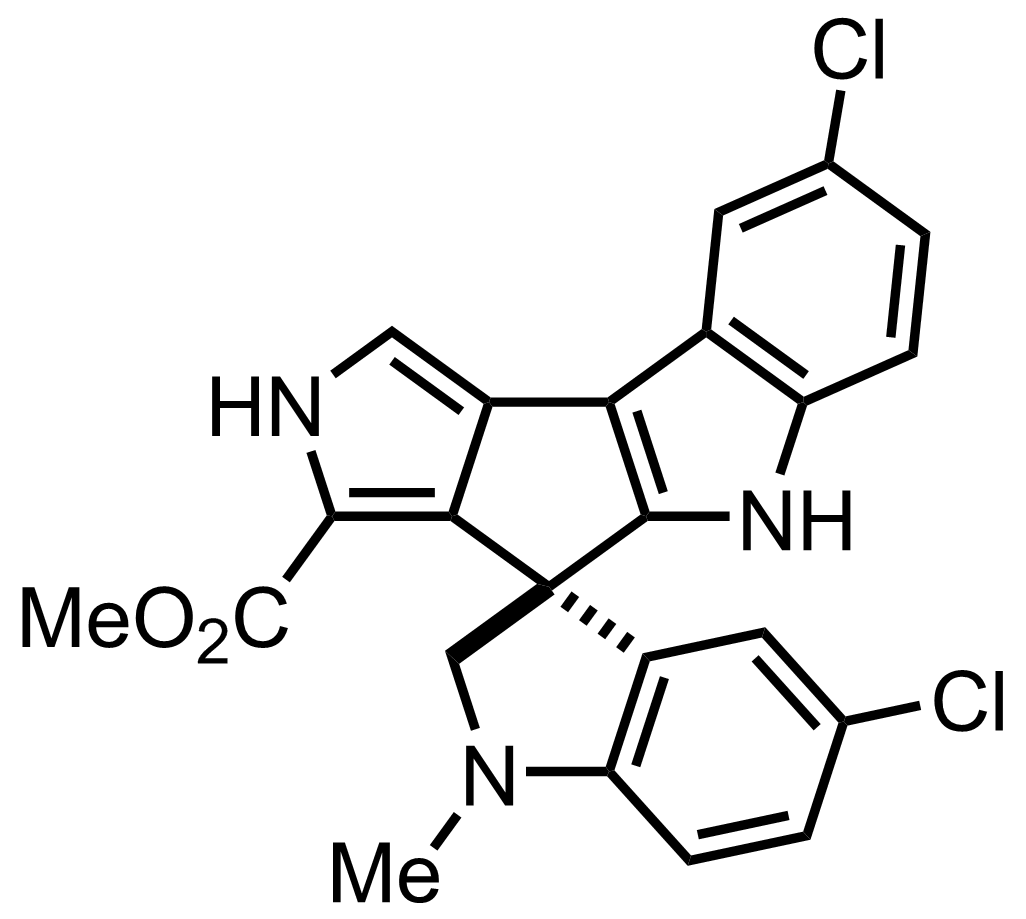
C23H17Cl2N3O2
| Principal investigator | Jonathan Sperry |
|---|---|
| Publication year | 2016 |
| Synthesis type | Total |
| Number of steps | 14 (linear) |
| References |
Part 1 of 1
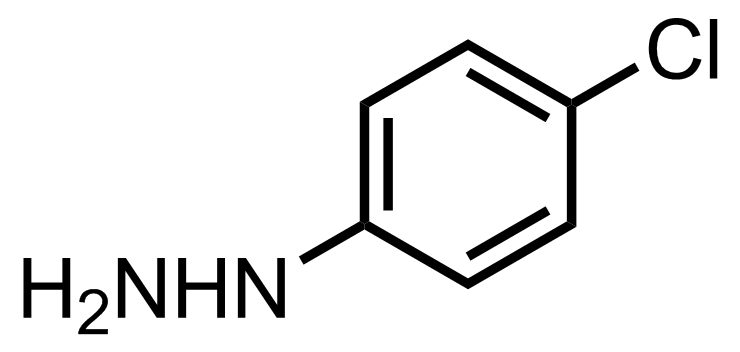
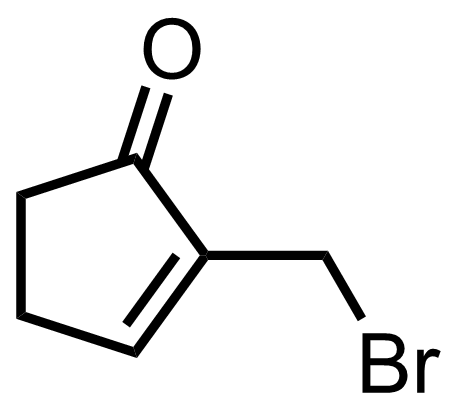 +
+
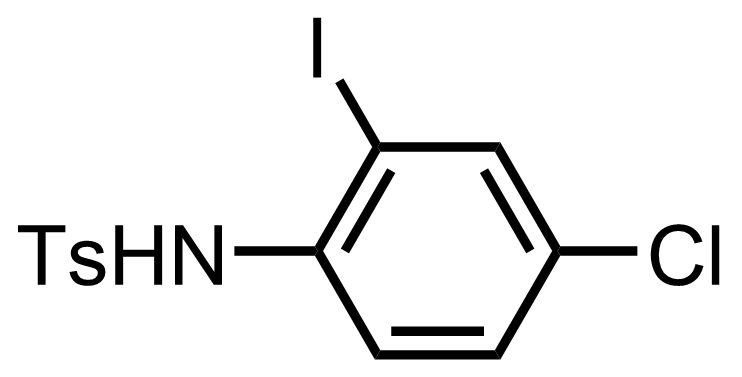




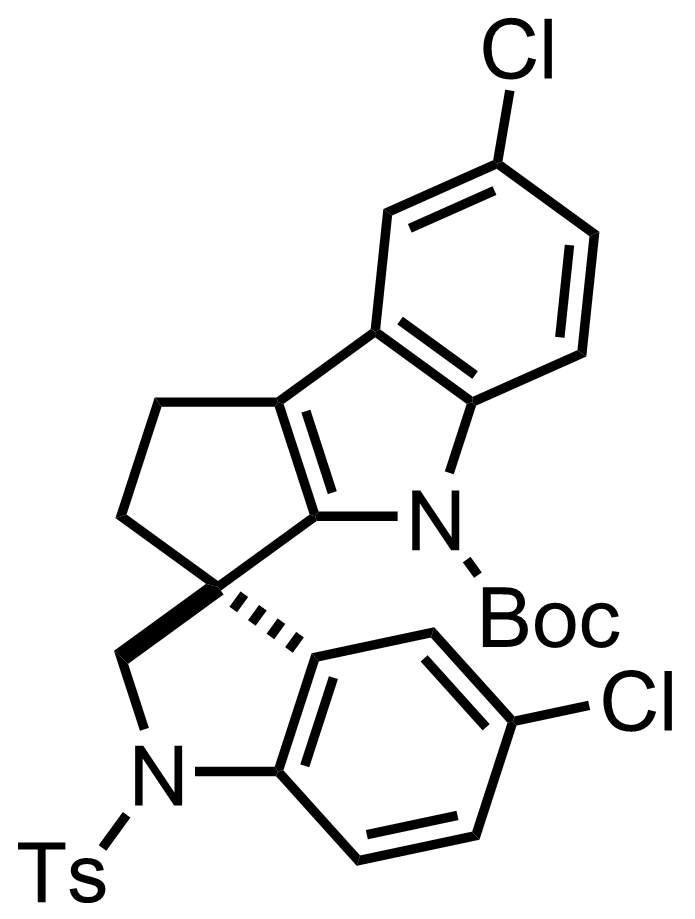
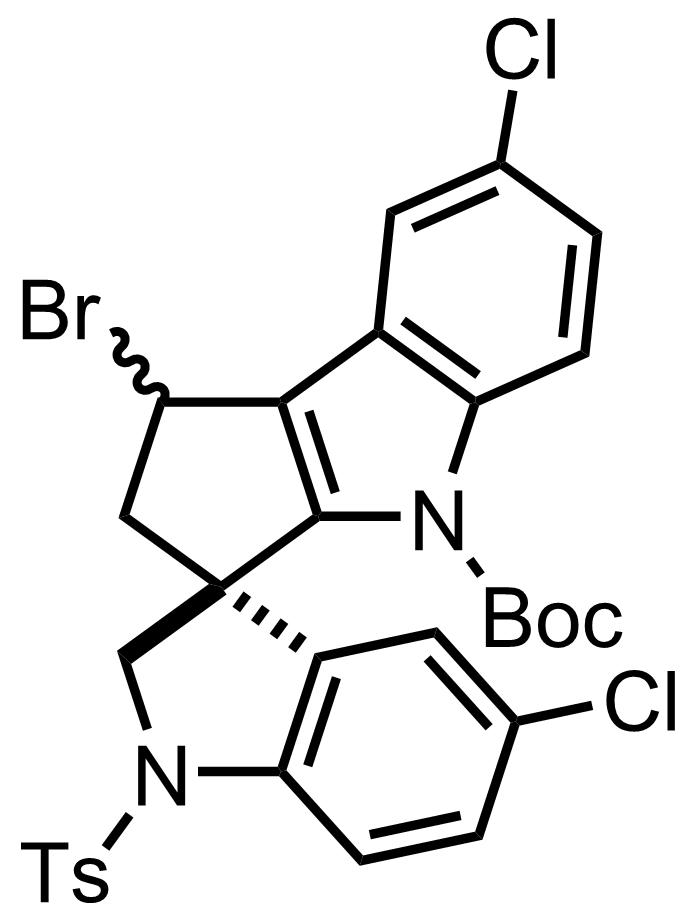
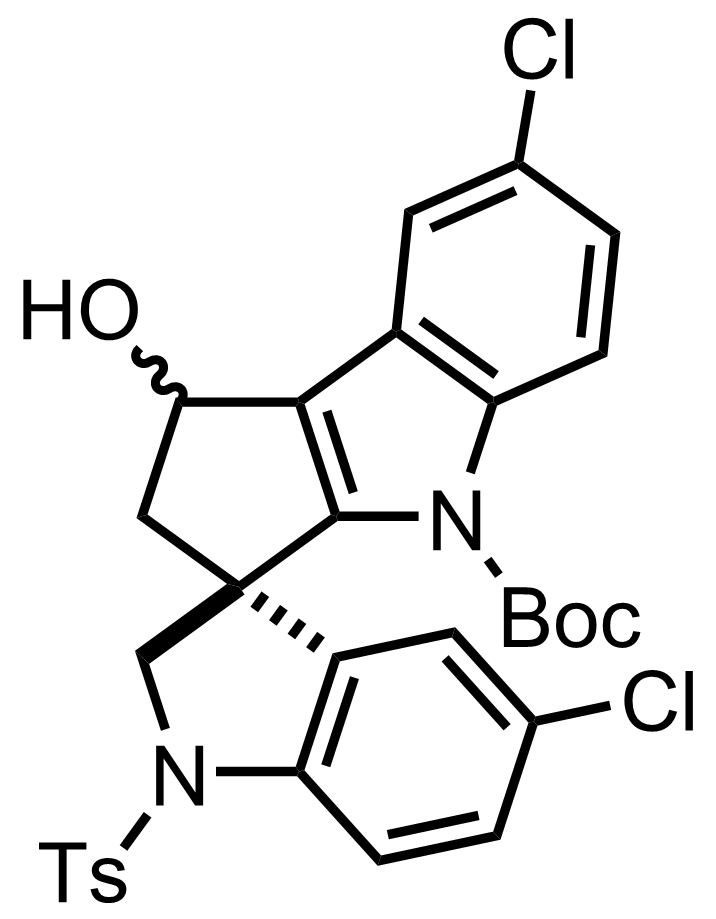

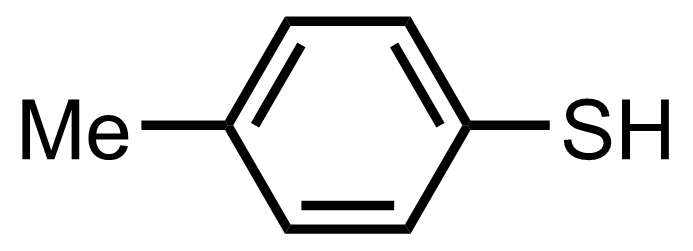
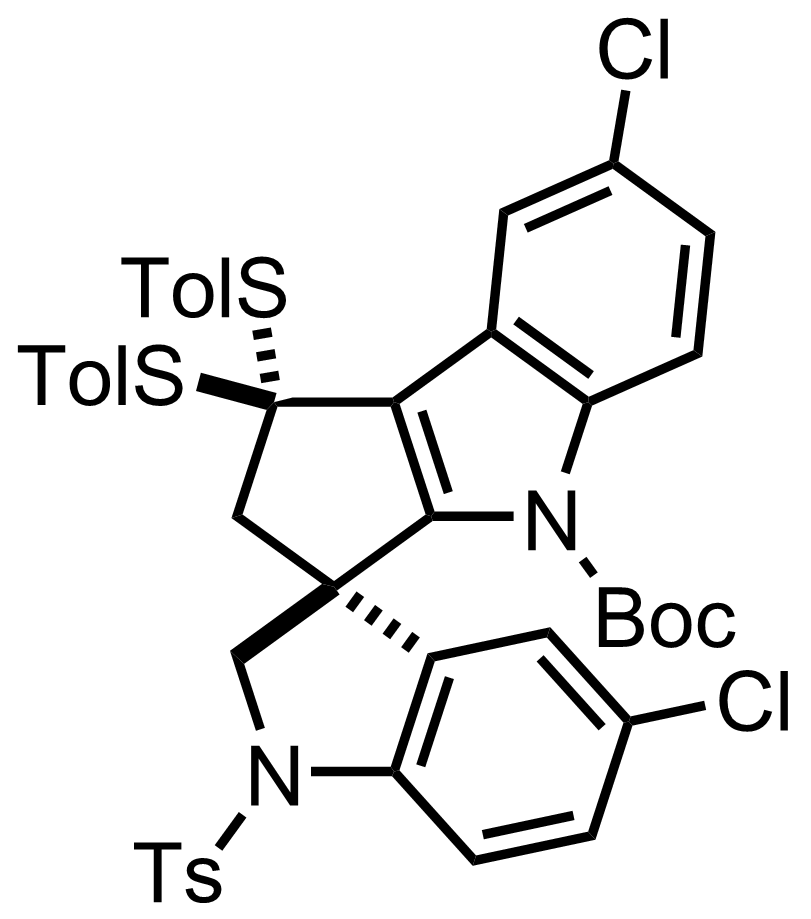
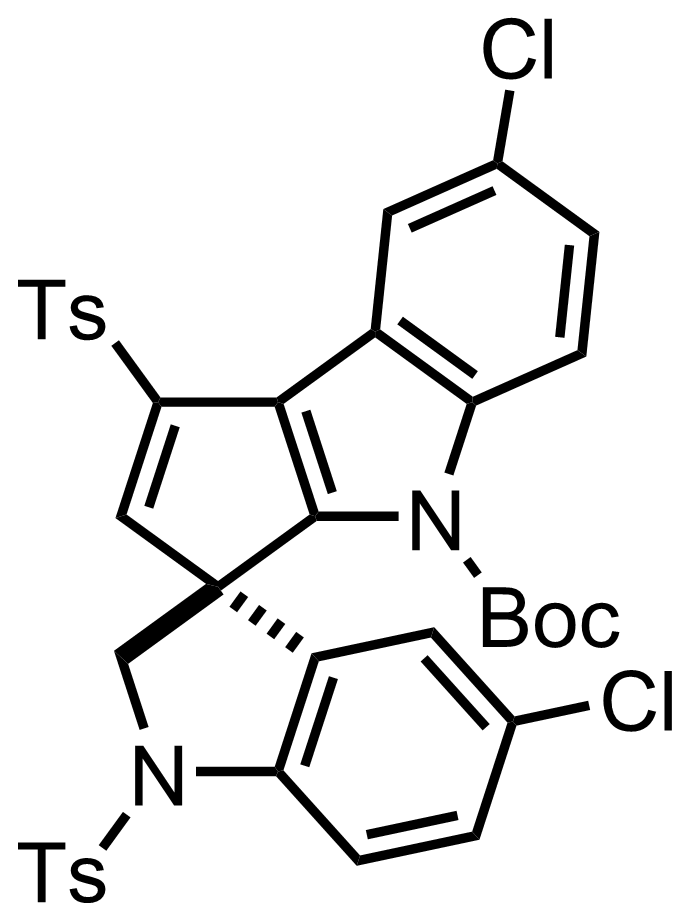
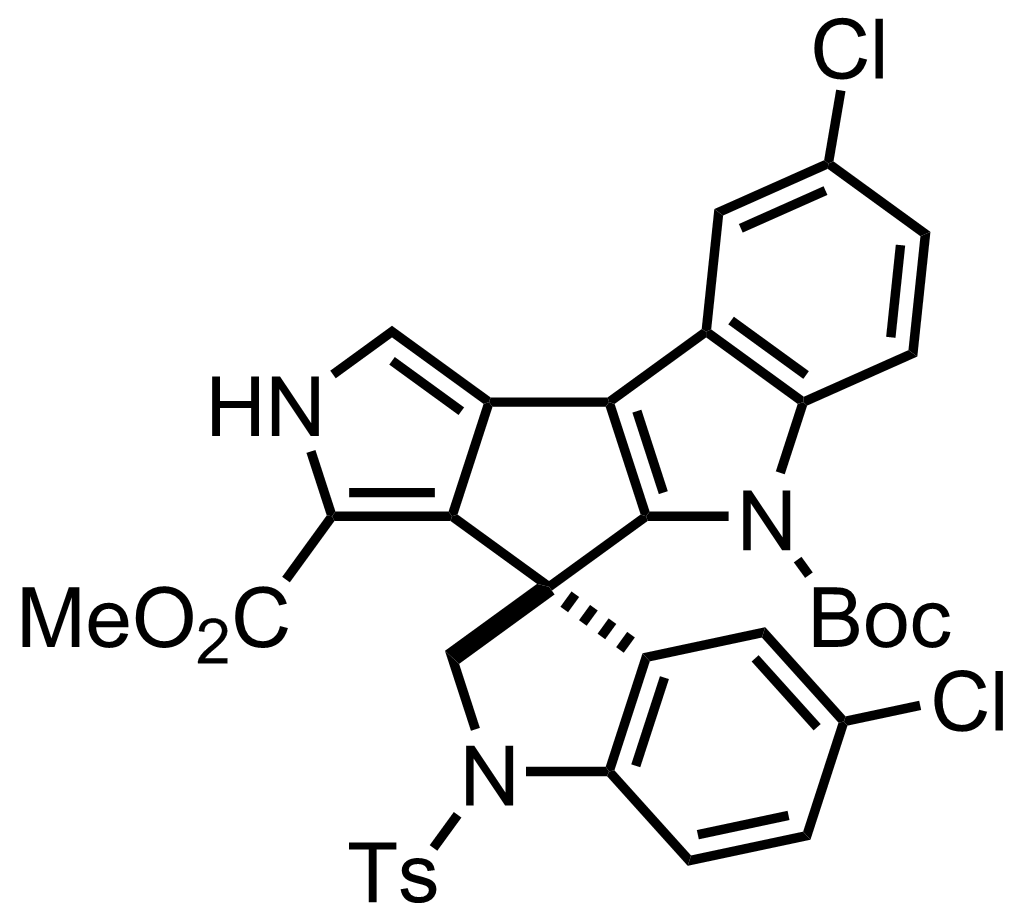
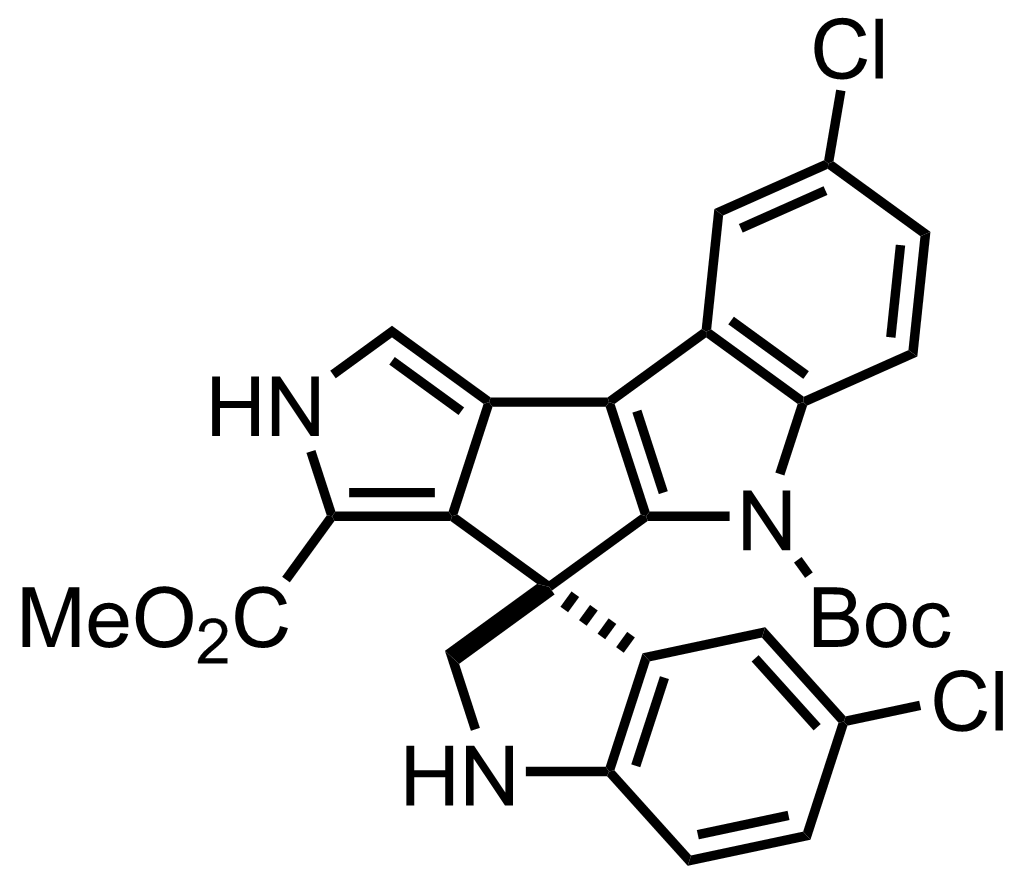
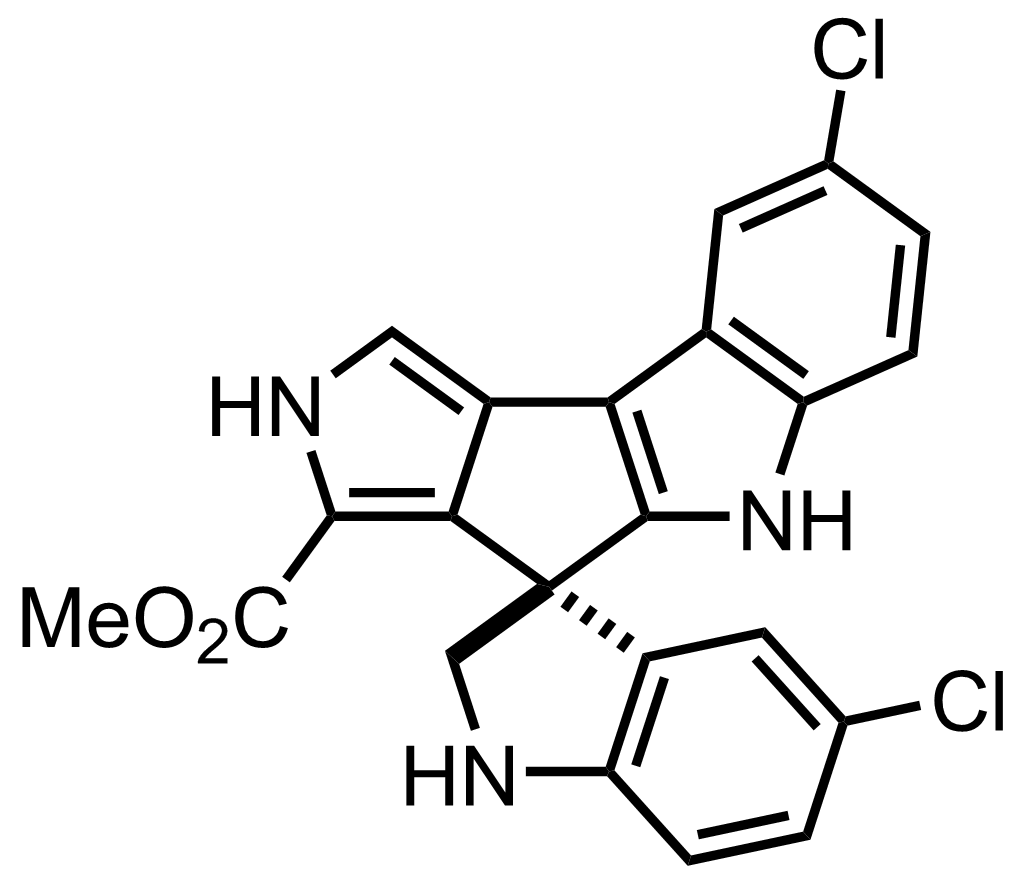
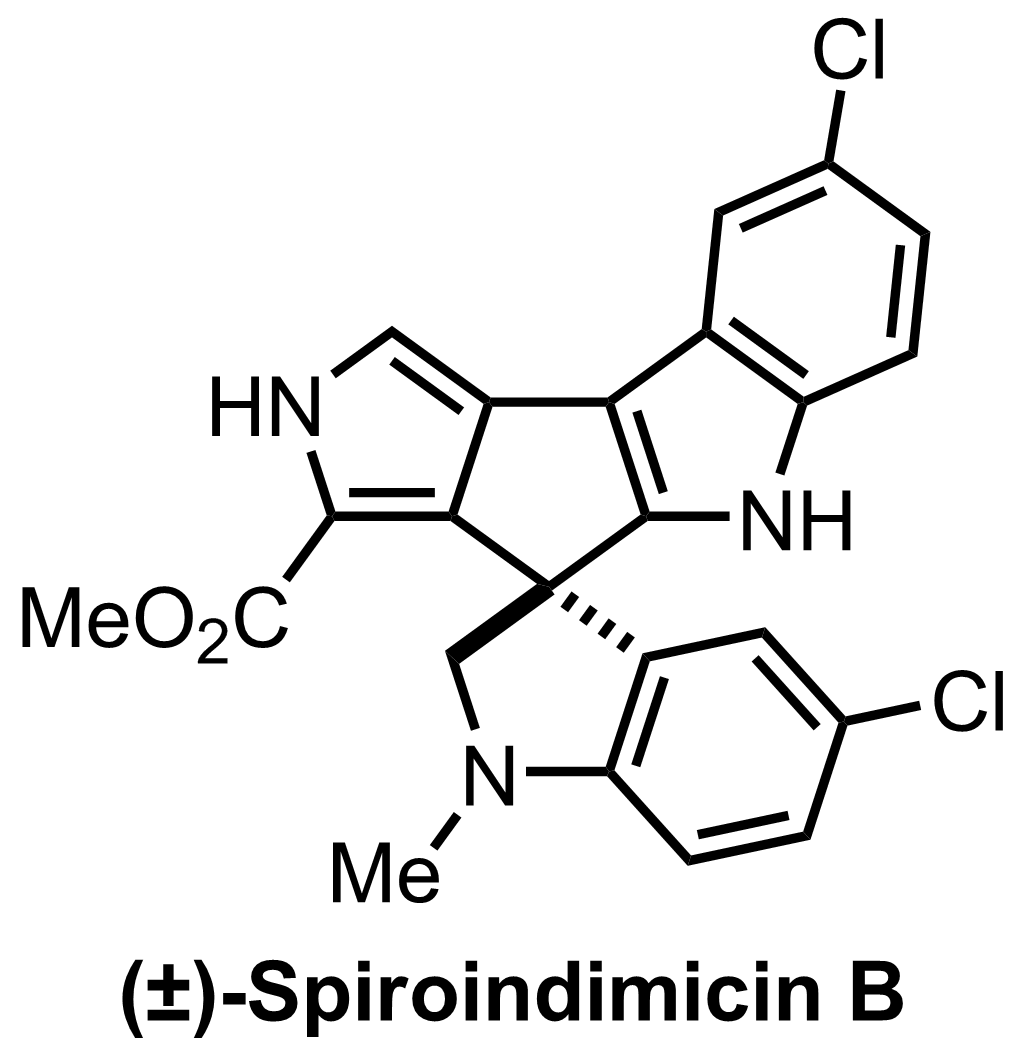
 +
+

NaH,
n-Bu4N+ I-
THF
0 °C to RT, 3.5 h, 85%


H2,
Pd/C
1 atm
EtOAc
RT, 60 min, 99%


DMAP,
Boc2O
THF
RT, 60 min, 96%


NaHCO3
H2O, THF
RT, 60 min

MnO2
CH2Cl2
RT, 20 h, 69% (3 steps)


Et3N,
TiCl4
THF
RT, 2 h, 78%

K2CO3,
mCPBA
CH2Cl2
0 °C to Reflux, 25 h, 39%
"Also isolated was a sulfoxide (31% yield) which was oxidized to the desired product."


Na.Naphthalene
THF
-78 °C

AlCl3
CH2Cl2
0 °C to RT, 35 min, 67% (2 steps)

AcOH,
CH2O,
NaBH3CN
H2O, MeOH
RT, 15 min, 92%